ISO 27701 for Healthcare Privacy Compliance
Post Summary
ISO 27701:2025 is a global standard designed to help healthcare organizations manage privacy risks involving sensitive patient data. Unlike its predecessor, it operates as a standalone framework, eliminating the need for ISO 27001 certification. This makes it accessible for healthcare providers, including small clinics and startups, to comply with privacy laws like HIPAA, GDPR, and CCPA. Key updates include guidance on AI, IoT, and cloud technologies, which are now central to modern healthcare.
Key Takeaways:
- Simplified Compliance: Consolidates privacy requirements for multiple regulations into one framework.
- Focus on Emerging Tech: Addresses risks tied to AI, cloud-based systems, and third-party IoT medical devices.
- Standalone Certification: No need for ISO 27001, reducing barriers for smaller organizations.
- Leadership Accountability: Strengthened oversight by management for privacy governance.
- Streamlined Audits: Reduces costs and time by integrating multiple privacy audits into one.
For healthcare providers, ISO 27701 certification demonstrates a commitment to safeguarding patient data while reducing regulatory complexity and operational risks.
How ISO/IEC 27701:2025 Privacy Information Management System Redefines Digital Trust Privacy webinar
sbb-itb-535baee
Core Requirements of ISO 27701 for Healthcare Organizations
ISO 27701:2025 lays out specific practices to enhance healthcare data privacy by implementing a Privacy Information Management System (PIMS). This standard emphasizes building, maintaining, and improving privacy protocols tailored for PII controllers and processors. For healthcare organizations managing Protected Health Information (PHI), this translates to embedding privacy management into everyday operations.
A key update in the 2025 edition is the concept of independent scoping, which allows organizations to focus their PIMS on specific areas, like marketing, without tying it to the larger Information Security Management System (ISMS) [2].
Leadership commitment is critical. Organizations need to assign clear responsibilities, such as appointing Risk Assessment Owners or DPIA Coordinators, particularly for high-risk processing tasks. Trust is also a major factor - 75% of consumers say they wouldn’t buy from a company they don’t trust with their data [2].
Let’s break down how to create, integrate, and evaluate an effective PIMS.
Building a Privacy Information Management System (PIMS)
The first step in establishing a PIMS is a gap analysis. This involves identifying where current data handling practices fall short of ISO 27701:2025 requirements. Healthcare organizations should document the personal data they collect, where it’s stored, its purpose, and who it’s shared with. Instead of static documentation, the focus should be on creating a dynamic system that tracks PII throughout its lifecycle.
For example, a hospital can map data flows from patient admission to record archiving. Non-clinical departments like HR (managing employee health records) or marketing (using patient data for outreach) also fall under the PIMS framework.
Integrating privacy management into organizational goals is essential. Assign specific roles to ensure accountability - don’t let privacy responsibilities scatter across legal, security, and operations teams. Turn privacy tasks into actionable, scheduled activities, such as training sessions, policy updates, and third-party risk assessments, rather than limiting them to annual audits.
Privacy Impact Assessments (PIAs) should be embedded into every processing stage. For instance, before launching a new telehealth app or diagnostic tool, a PIA can uncover privacy risks early, ensuring data processing remains transparent and manageable.
| Component | Healthcare Application (PHI) |
|---|---|
| PII Controller | Hospitals or clinics determining how patient data is used for treatment. |
| PII Processor | Third-party billing or IT vendors handling PHI on behalf of the hospital. |
| Privacy Impact Assessment (PIA) | Evaluating the risk of a new telehealth app before deployment. |
| Privacy by Design | Designing a new Electronic Health Record (EHR) system to collect only necessary patient data. |
| Independent Scoping | Applying PIMS to marketing teams using patient contact information for outreach. |
Privacy by Design and Default in Healthcare
Privacy by Design ensures privacy principles are built into systems, workflows, and applications from the start. ISO 27701 transforms this concept into structured processes with clear responsibilities and evidence-based decisions.
For healthcare organizations, this means conducting Data Protection Impact Assessments (DPIAs) whenever new tools - like medical apps, wearable devices, or telehealth platforms - are introduced. DPIAs are particularly important for high-risk activities, helping ensure privacy is a core consideration at every stage. For example, when developing an Electronic Health Record (EHR) system, data minimization should guide its design, collecting only the necessary information for clinical purposes.
Strict access controls and encryption of PHI, as outlined in GDPR Article 32, are vital. Healthcare organizations must also document the legal basis for processing data and establish retention limits.
"ISO 27701 is about making privacy operational."
– Deiterate
Fragmented privacy records can create vulnerabilities. Disjointed systems for policies, evidence, and risk registers can lead to overconfidence and slower response times during breaches. Organizations adopting ISO 27701 have reported a 60–80% improvement in response times to Data Subject Access Requests (DSARs) through automated discovery [3].
By following these principles, healthcare organizations can ensure privacy is thoroughly integrated into their operations.
Conducting Privacy Risk Assessments
Under ISO 27701, privacy risk assessments require healthcare organizations to catalog all PII processing activities and identify potential threats, such as unauthorized access, accidental loss, or unlawful disclosure. The standard demands a dual-impact analysis, evaluating both the likelihood of a risk and its consequences for the organization and affected individuals.
Given the sensitive nature of health data, risk treatment must be particularly rigorous. Organizations should adapt their risk scales to automatically flag "High" or "Critical" ratings for sensitive patient information. High-priority risks can be addressed by selecting controls from Annex A, documenting the reasoning behind each control, and assessing any remaining risks.
Assigning clear accountability is key. Roles like Risk Assessment Owners and DPIA Coordinators ensure that privacy risks are managed effectively across clinical departments. Additionally, ISO 27701’s risk framework can align with HIPAA or GDPR requirements, simplifying compliance and reducing the workload compared to handling multiple audits independently.
Key Updates in ISO 27701:2025 for Healthcare
ISO 27701:2019 vs 2025 Key Differences for Healthcare Privacy Compliance
The 2025 edition of ISO 27701 introduces updates tailored to modern healthcare challenges. Unlike the 2019 version, which extended ISO 27001, the new standard operates as a standalone Privacy Information Management System (PIMS). This change eliminates the need for prior ISO 27001 certification, making privacy certification more accessible to healthcare providers focused on data protection.
The updated standard also directly addresses technologies now common in healthcare, such as AI-driven diagnostics, automated decision-making, and IoT-enabled patient monitoring. For instance, hospitals using AI analytics for patient profiling or predictive care must now conduct specific risk assessments under the revised framework.
"Privacy is no longer subordinate to information security. It is now an equal governance discipline."
– Brian Kline, COO, Neutral Partners [4]
In addition, leadership accountability has been bolstered. Clause 5 requires top management to actively oversee privacy governance, ensuring it is treated as a core business function rather than being delegated to technical teams. This shift aligns privacy with other critical areas of governance, demanding the necessary resources and attention.
For organizations preparing for ISO 27701:2025, readiness typically requires 8 to 16 weeks, depending on factors like size and governance maturity [4]. Healthcare CISOs should prioritize updating Data Protection Impact Assessment (DPIA) inventories and revising vendor agreements to align with the updated definitions of processor and controller roles.
Stand-Alone PIMS Standard
One of the most notable changes in ISO 27701:2025 is its independence from ISO 27001. This is particularly beneficial for smaller clinics and health-tech startups, which can now achieve privacy certification without implementing a full Information Security Management System (ISMS). This flexibility is especially useful for organizations needing to demonstrate privacy compliance without the broader scope of ISO 27001.
The 2025 edition adopts the Annex SL high-level structure, aligning it with other major ISO standards like ISO 9001 and ISO 27001:2022. This harmonization simplifies integration for organizations already certified in other ISO frameworks, making it easier to incorporate privacy management into existing governance processes.
For North American healthcare providers, pairing SOC 2 for security with ISO 27701:2025 for privacy is becoming increasingly popular [4]. This approach avoids duplicating a full ISO 27001 audit while providing comprehensive assurances to patients, payers, and business partners. Organizations can choose between an independent privacy audit or an integrated assessment with their existing certifications, depending on their operational needs.
Updated Privacy Risk Management
The 2025 edition introduces specific requirements for managing risks tied to emerging technologies that are now integral to healthcare. These include detailed guidance on AI-driven analytics, automated decision-making systems, cloud-based processing, and cross-border data transfers.
For example, a hospital utilizing AI to predict patient readmission rates must now assess not only the tool's clinical accuracy but also the privacy risks of automated profiling. Organizations are required to document how these systems make decisions, what data they rely on, and how patients can understand or challenge automated outcomes.
New controls also emphasize data masking, Data Leakage Prevention (DLP), and ICT readiness for business continuity [5]. These updates are especially relevant for healthcare systems using cloud-based Electronic Health Records (EHR) or managing data across multiple jurisdictions. Organizations must now implement privacy controls for data residency and cross-border transfers.
Additionally, the updated standard mandates designated risk owners and clear criteria for privacy assessments. This means assigning specific roles, such as DPIA Coordinators or Privacy Officers, to oversee high-risk activities like telehealth platforms or wearable device integrations. Healthcare organizations should update their risk registers to reflect these new technology-specific threats and document treatment plans accordingly.
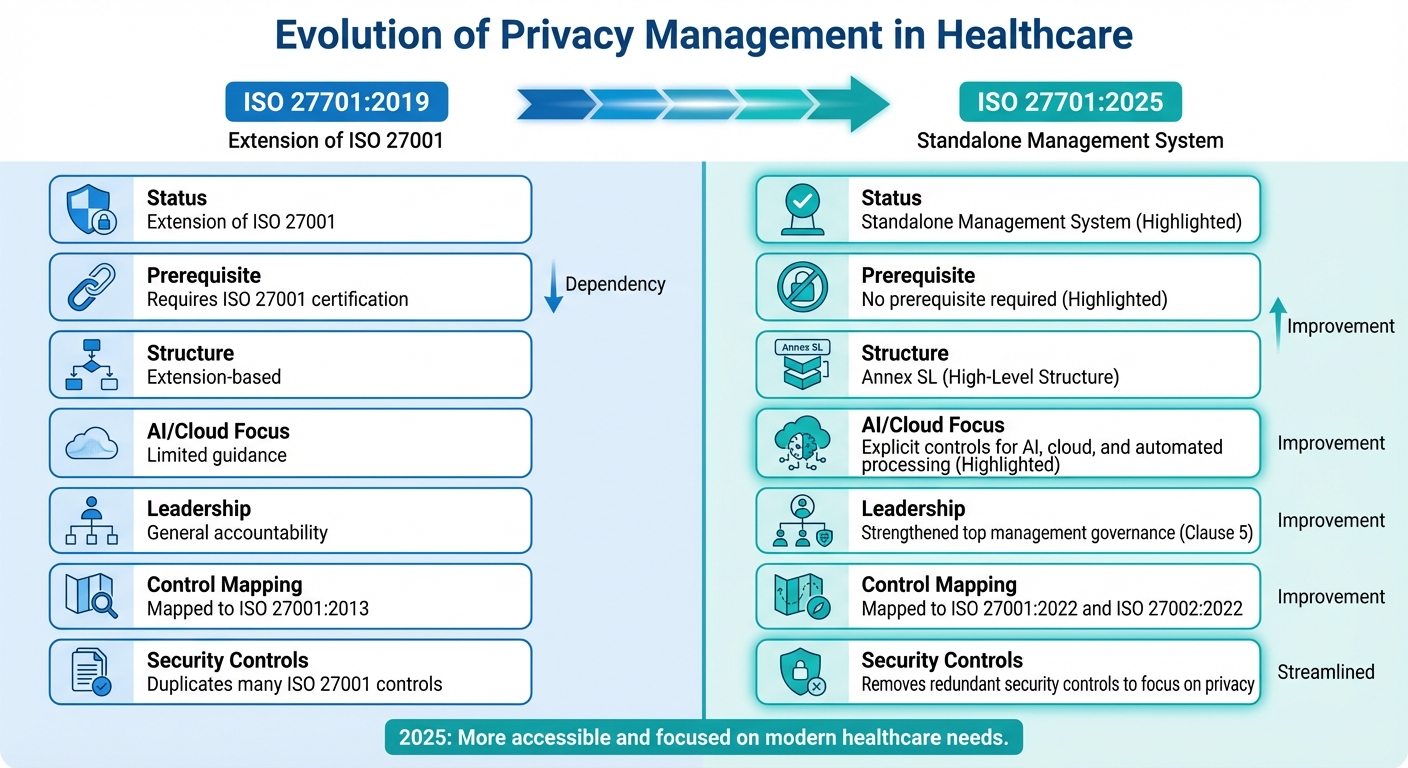
ISO 27701:2019 vs. 2025 Comparison
Understanding the changes between the two editions is essential for healthcare organizations planning their transition. The table below highlights key differences:
| Feature | ISO 27701:2019 | ISO 27701:2025 |
|---|---|---|
| Status | Extension of ISO 27001 | Standalone Management System |
| Prerequisite | Requires ISO 27001 certification | No prerequisite required |
| Structure | Extension-based | Annex SL (High-Level Structure) |
| AI/Cloud Focus | Limited guidance | Explicit controls for AI, cloud, and automated processing |
| Leadership | General accountability | Strengthened top management governance (Clause 5) |
| Control Mapping | Mapped to ISO 27001:2013 | Mapped to ISO 27001:2022 and ISO 27002:2022 |
| Security Controls | Duplicates many ISO 27001 controls | Removes redundant security controls to focus on privacy |
The 2025 edition sharpens its focus on privacy by eliminating redundant security controls and adding specific guidance for AI, cloud, and automated processing [5]. This streamlined approach reduces the audit burden while clarifying the distinct roles of privacy and security management.
Healthcare organizations transitioning from the 2019 edition should conduct a gap analysis to compare their current practices with the new requirements. Special attention should be given to controls for AI, automated processing, and cross-border data flows, as these represent the most substantial updates. Vendor agreements should also be updated to reflect the refined roles of processors and controllers, especially for cloud-based subcontractors handling Protected Health Information (PHI). These changes highlight the standard's tighter focus on privacy and its alignment with the needs of modern healthcare.
Benefits of ISO 27701 Certification for Healthcare Organizations
ISO 27701 certification simplifies compliance by consolidating various privacy regulations into a single Privacy Information Management System (PIMS). This approach not only reduces audit expenses but also minimizes risks. By implementing standardized controls that meet regulatory expectations, healthcare organizations can avoid the challenges of managing fragmented compliance efforts. The certification also provides the documentation that regulators typically require, which can be streamlined using automated security questionnaires.
The financial impact of non-compliance can be severe, with potential fines reaching $18 million or 4% of global turnover [1]. ISO 27701 helps mitigate these risks by adopting a proactive, risk-based strategy to address emerging threats to patient privacy. Furthermore, organizations with this certification may be viewed more favorably by regulators, such as the UK Information Commissioner’s Office (ICO), during investigations into data breaches [1].
Improved Trust and Compliance
Certification offers independent validation of privacy practices, building confidence among patients, business associates, and payers. Mike Jennings, IMS Manager at ISMS.online, highlights this benefit:
"ISO 27701 is an impressive way of demonstrating to consumers, external organisations and internal stakeholders, that mechanisms are in place to keep data safe and to comply with GDPR and other privacy laws" [1].
For healthcare organizations operating across different regions, ISO 27701’s neutral design simplifies compliance by enabling a single set of controls to meet various local regulations. It also clearly defines the roles of PII controllers and processors, which is essential in complex healthcare ecosystems involving cloud EHR vendors, medical device manufacturers, and billing processors [1][7]. This clarity not only enhances trust but also makes audits more efficient.
Easier Audits and Third-Party Agreements
ISO 27701 streamlines the audit process, allowing internal and external auditors to evaluate compliance with multiple regulations in a single cycle [1][8]. This integrated approach is more cost-effective than conducting separate audits for frameworks like HIPAA and state privacy laws. Karen C., a reviewer, shared her experience:
"Makes external audits a breeze and links all aspects of your ISMS together seamlessly" [1].
Certification also speeds up vendor agreements by providing standardized, audited evidence that simplifies data-sharing processes. With triennial audits, organizations can ensure ongoing compliance without constant re-certification efforts.
Certification Benefits and Implementation Trade-Offs
Healthcare organizations must weigh the benefits of certification against the resources required for implementation. Here’s a closer look:
| Benefits | Challenges |
|---|---|
| Risk Reduction: Identifies and mitigates privacy risks to patient data [1]. | Resource Investment: Requires dedicated roles, such as Data Protection Officers and Privacy Analysts [1]. |
| Trust Building: Provides independent validation of privacy practices for stakeholders [6][7]. | Audit Costs: Involves preparation and external registrar fees [1]. |
| Simplified Audits: Covers multiple regulatory requirements in one cycle [1]. | Complexity: Integrating privacy controls into existing workflows [1]. |
| Faster Vendor Agreements: Streamlines data-sharing processes [1][7]. | Maintenance: Requires ongoing audits and continuous improvement efforts [1]. |
The cost of certification varies depending on the size of the organization. For instance, healthcare organizations with 126–425 employees typically spend $10,500–$11,700, while larger systems with 1,551–2,025 employees may invest around $17,500 [1]. Organizations already certified to ISO 27001 can reduce expenses by implementing both standards simultaneously [1][6].
The built-in Plan, Do, Check, Act (PDCA) cycle in ISO 27701 ensures that the PIMS adapts to new healthcare technology risks, such as AI-driven diagnostics and IoT-enabled patient monitoring. By focusing on ongoing improvement, organizations can address privacy threats proactively rather than reacting to incidents after they occur [1]. This approach equips healthcare organizations to stay ahead in managing evolving data risks effectively.
Implementing ISO 27701 in Healthcare with Censinet RiskOps™

Healthcare organizations aiming to adopt ISO 27701 can simplify the process with platforms like Censinet RiskOps™, which automates privacy governance workflows and reduces manual effort for certification. This platform centralizes risk assessments, evidence collection, and HIPAA-compliant vendor risk management evaluations into a single system, helping organizations meet the privacy risk management standards outlined in the updated 2025 guidelines. Let’s explore how RiskOps™ supports gap analysis and real-time risk monitoring.
Gap Analysis and Risk Management
Censinet RiskOps™ helps healthcare providers pinpoint compliance gaps by comparing current privacy controls against ISO 27701’s requirements. With its risk assessment tools, organizations can evaluate privacy risks across key areas like patient data, PHI, clinical applications, and medical devices. By prioritizing remediation efforts based on the severity of these gaps, teams can focus on implementing the most critical privacy controls first.
The platform’s command center provides real-time updates on privacy risks, giving Data Protection Officers and Privacy Analysts a clear view of progress toward ISO 27701 compliance. Additionally, organizations can document their Privacy Information Management System (PIMS) directly in RiskOps™, creating a detailed audit trail to showcase compliance with the standard’s operational requirements.
Automated Privacy Governance via Censinet AITM
Censinet AITM speeds up evidence collection and documentation by automating key steps in privacy governance. Its AI-powered tools summarize vendor evidence, track integration details, and identify risks from fourth-party vendors - essential for meeting ISO 27701 requirements for both PII controllers and processors.
This automated process routes privacy findings to the appropriate stakeholders for review and approval, ensuring accountability across Governance, Risk, and Compliance (GRC) teams. It also supports the continuous improvement cycle outlined in ISO 27701’s Plan, Do, Check, Act (PDCA) framework.
Preparing for Certification with Censinet Connect™
Building on its automation capabilities, Censinet Connect™ simplifies vendor risk assessments, which are crucial for healthcare organizations managing complex networks of cloud EHR systems, medical device manufacturers, and billing processors. The platform provides standardized, audited evidence that can be shared with certification bodies during audits.
Using Connect™, organizations can demonstrate their approach to managing data-sharing agreements and third-party privacy controls in line with ISO 27701’s requirements. This streamlined process not only reduces the time and cost of external audits but also eliminates the need for last-minute documentation efforts, allowing healthcare providers to maintain a strong PIMS and focus on operational excellence.
Conclusion
ISO 27701 brings together scattered, regulation-specific privacy efforts into a cohesive framework that covers the entire lifecycle of Personally Identifiable Information (PII). By merging your Information Security Management System (ISMS) with a Privacy Information Management System (PIMS), you create a solid foundation for meeting privacy standards like HIPAA and GDPR while cutting down on audit fatigue and associated costs.
The 2025 update to ISO 27701 takes this framework a step further by positioning privacy as a fully developed, independent discipline with a streamlined certification process. For healthcare organizations, this means a chance to showcase global best practices through a single audit - a critical advantage in light of the serious consequences of non-compliance with data protection laws.
Tools like Censinet RiskOps™ make achieving certification easier by automating key processes such as gap analysis, privacy risk assessments, and vendor evaluations. Features like Censinet AITM, which automates evidence collection, and Censinet Connect™, which oversees third-party controls, allow healthcare providers to focus on operational efficiency while driving continuous improvement.
As Mike Jennings, IMS Manager at ISMS.online, aptly puts it:
"ISO 27701 supports effective privacy and information security while reducing risks" [1].
For healthcare organizations navigating complex supply chains and ever-changing regulations, combining ISO 27701 certification with automated governance tools creates a strong operational framework. This approach not only embeds best practices into daily workflows but also fosters trust among patients, partners, and regulators.
FAQs
What’s the fastest way to scope ISO 27701 to just one department?
To narrow the scope of ISO 27701 to just one department, center your efforts on tailoring the standard’s controls to that department’s specific operations. Begin by clearly outlining the department’s boundaries, identifying its data processing activities, and mapping out how data flows within its processes. From there, apply the relevant controls directly to these activities.
Create policies that are specific to the department’s needs, carry out a focused risk assessment for its operations, and set up monitoring systems to ensure compliance. This approach allows for effective privacy management within the department without needing to implement the standard across the entire organization.
How does ISO 27701:2025 address AI-based patient profiling and automated decisions?
ISO 27701:2025 introduces specific guidelines to tackle privacy concerns tied to AI. These include requirements for transparency and oversight, ensuring AI-driven processes are handled responsibly. This is especially crucial in healthcare, where tasks like patient profiling and automated decision-making demand careful management to protect sensitive information and maintain trust.
What evidence is required to pass an ISO 27701 audit in healthcare?
To successfully clear an ISO 27701 audit in the healthcare sector, organizations need to prove they have put in place robust measures for managing Personally Identifiable Information (PII). This involves maintaining detailed records of processing activities, ensuring privacy commitments to PII principals are met, conducting thorough risk assessments, and following the ISO 27002 guidelines tailored for PII controllers. The evidence provided must clearly align with the framework's standards, showcasing compliance with privacy and data governance requirements.


